- Email us: sales@msbdocs.com

Zydus Lifesciences is a global pharmaceutical company operating across highly regulated markets, managing quality, engineering, and regulatory documentation under FDA 21 CFR Part 11, ICH, and global GMP standards.
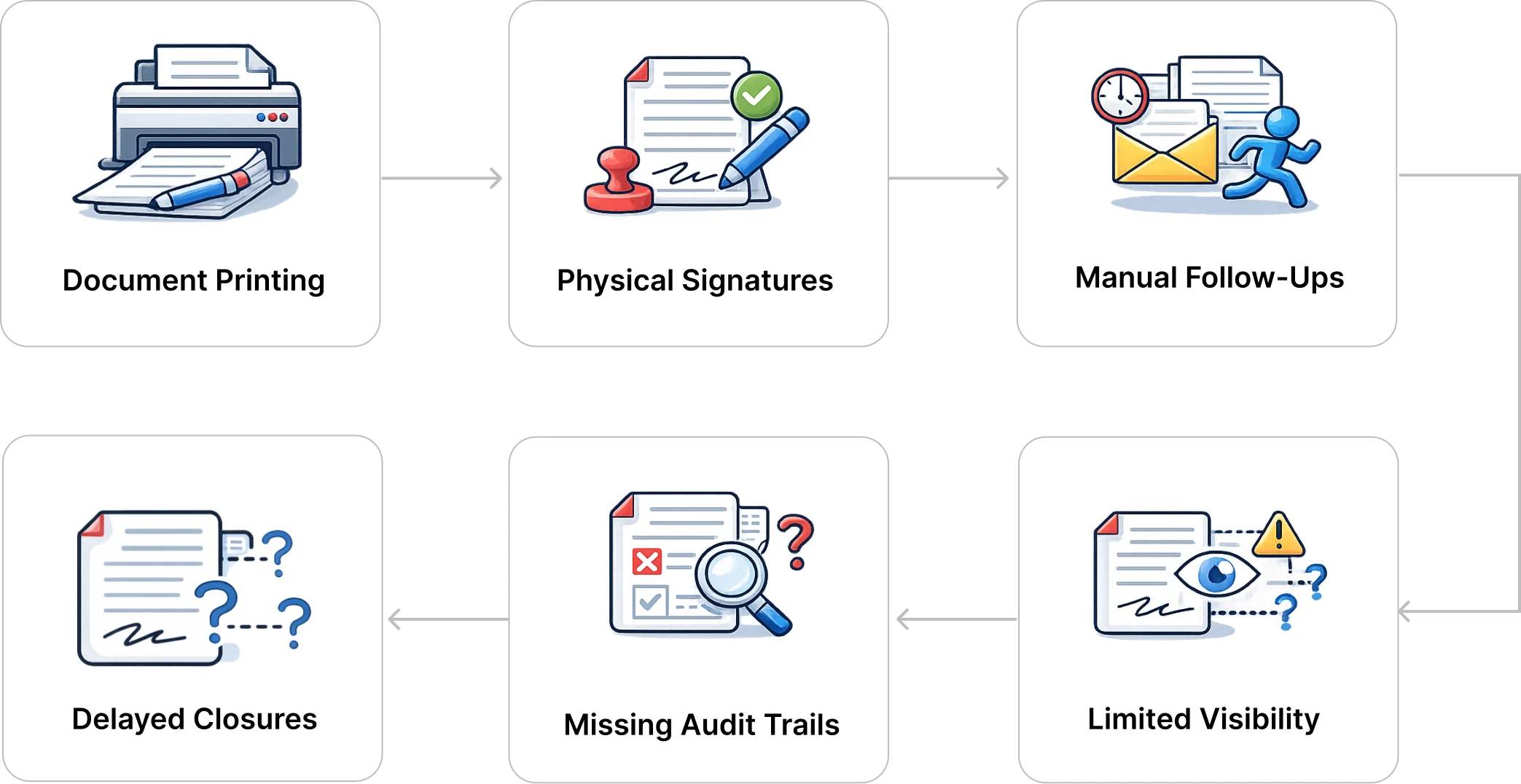
High-volume, compliance-critical documentation relied on manual and semi-digital approvals, leading to delays in investigations, CAPA closures, change controls, and audit readiness.
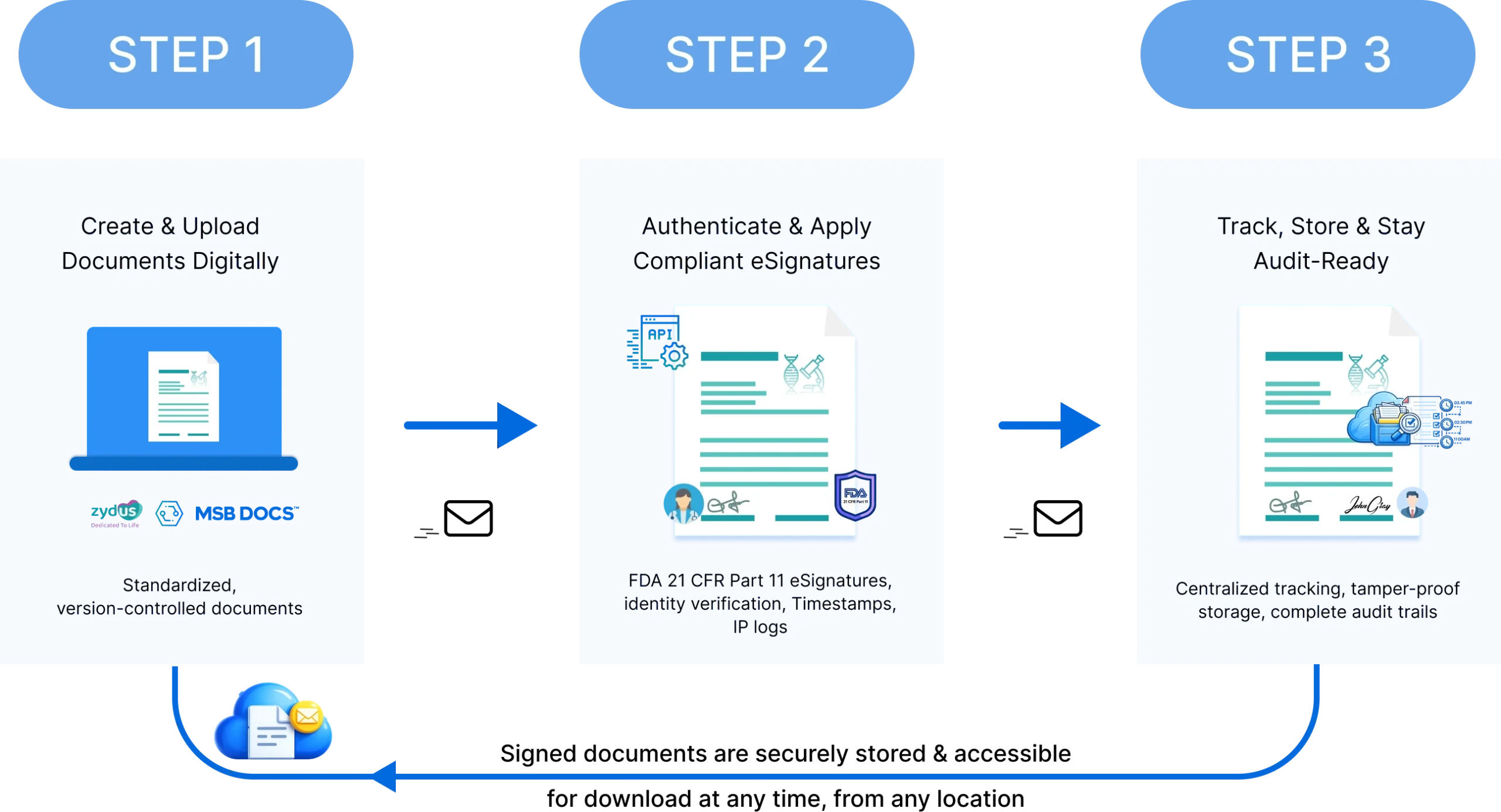
Zydus Lifesciences adopted MSB Docs eSignature to digitize and standardize the execution of regulated documents across departments.
After implementing MSB Docs, Zydus Lifesciences achieved measurable improvements across compliance, speed, and governance:
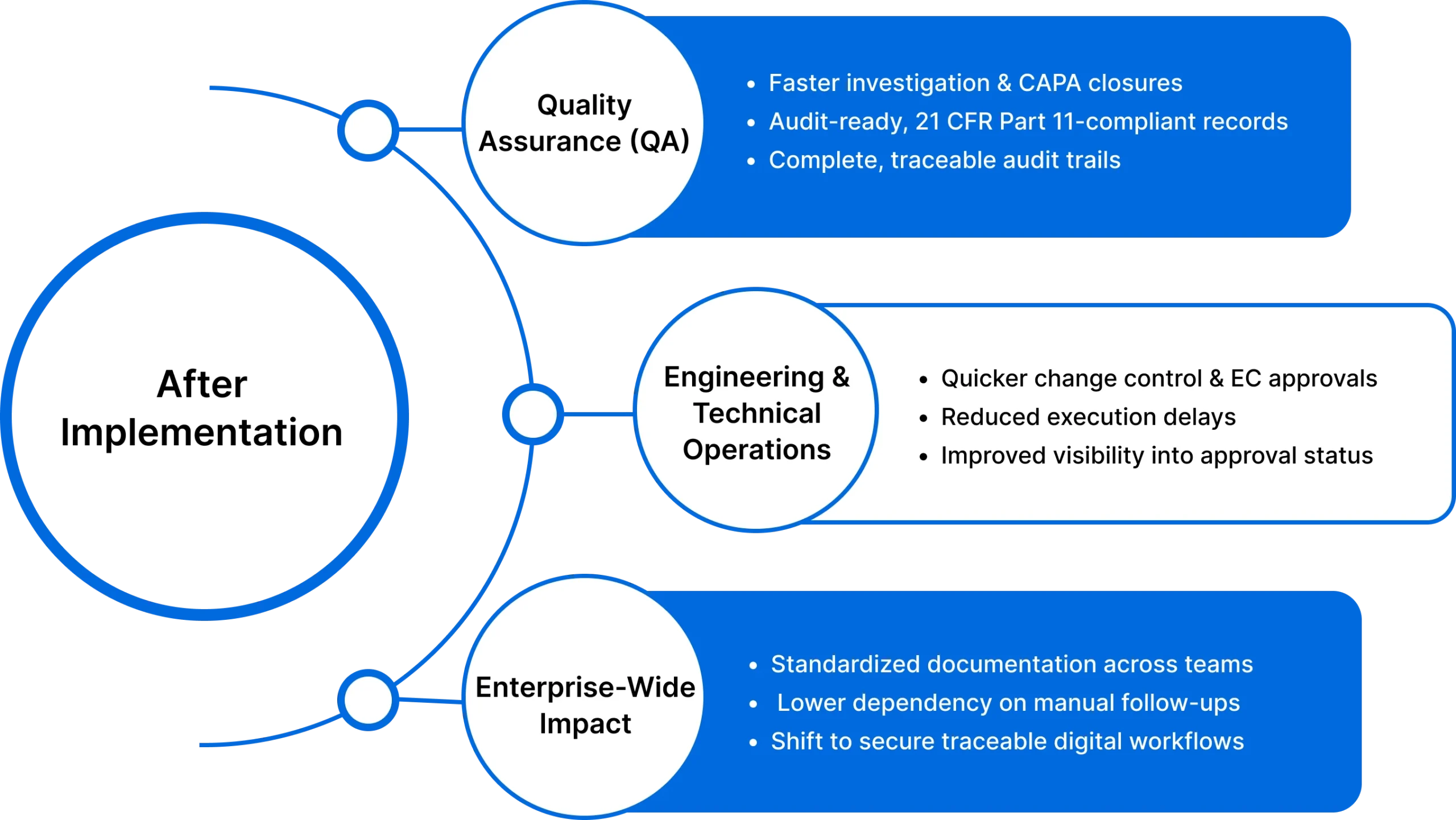
Reduced document turnaround time
Complete, audit-ready trails
Paper-free execution
MSB Docs eSignature helped Zydus Lifesciences accelerate and speed up approvals while strengthening compliance across regulated workflows. By adopting eSignatures as a compliance control, Zydus achieved secure, traceable, and audit-ready documentation. Life sciences organizations looking to reduce approval delays and improve inspection readiness can start their free trial of MSB Docs today and digitize with confidence.