
How Glenmark Simplifies Documentation
Signing with MSB Docs eSignature
About Glenmark Pharmaceuticals
Glenmark Pharmaceuticals Ltd., founded in 1977, is a global, research-driven pharmaceutical company with a strong presence in India and operations in 80+ countries. It develops branded generics, specialty, and OTC medicines, while prioritizing quality, regulatory compliance, and patient safety through continuous investment in digital quality and regulatory systems.
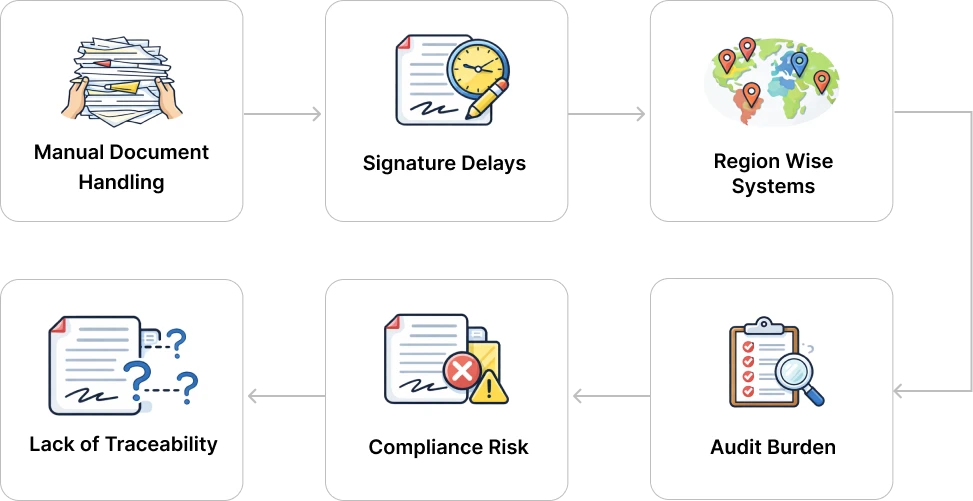
Business Challenge (Before Implementation)
Prior to implementation, Glenmark faced operational and compliance challenges common to large, regulated pharmaceutical enterprises:
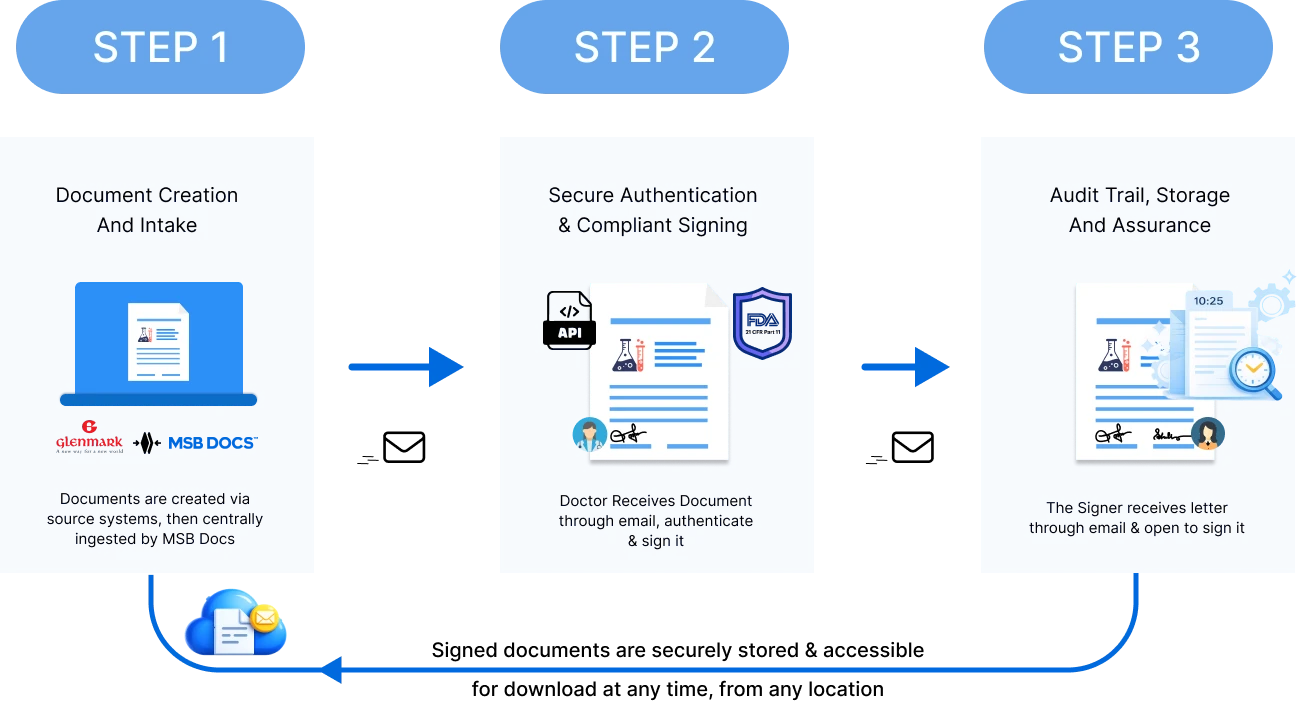
Solution Implemented
(MSB Docs eSignature – 21CFR Part 11)
Glenmark implemented MSB Docs’ 21 CFR Part 11–compliant eSignature solution with API integrations to digitize and standardize document signing across its global operations.
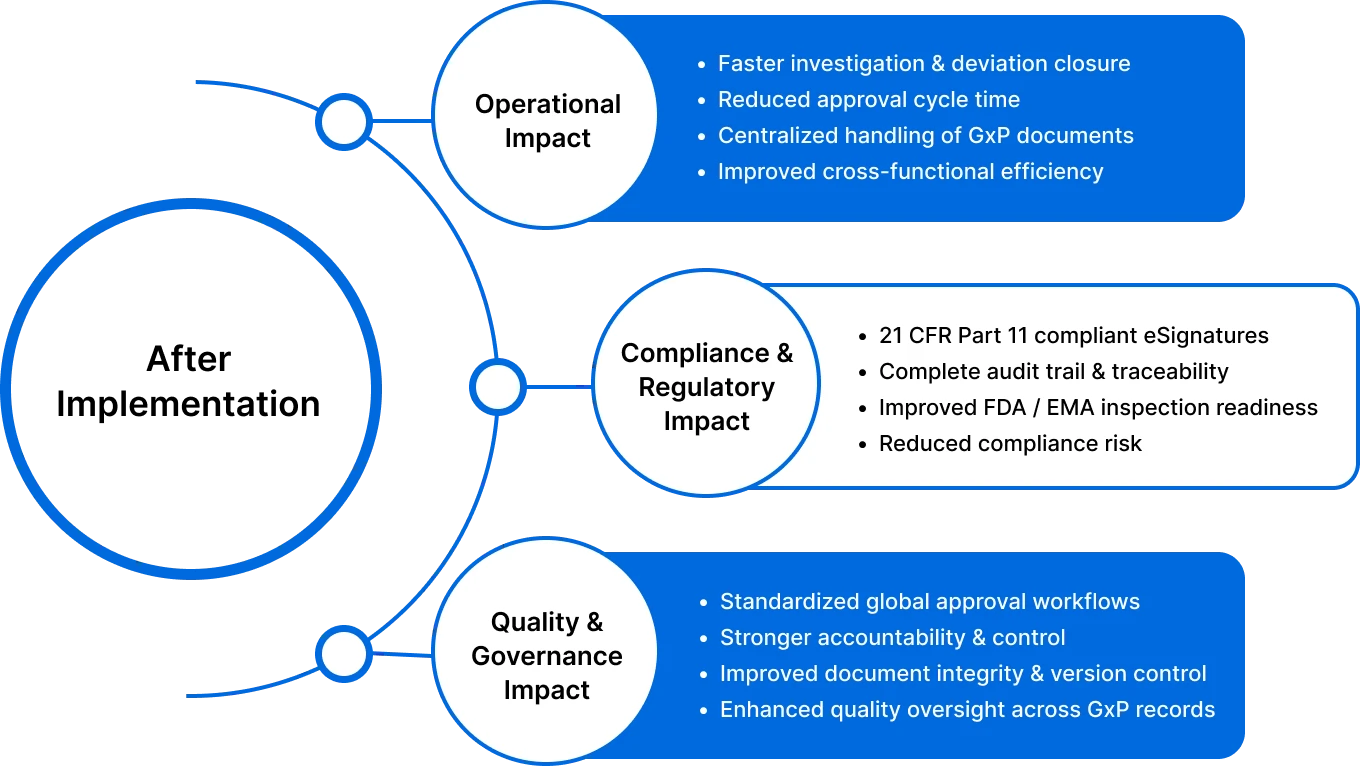
Glenmark Business Impact
Glenmark’s digital solutions speed up approvals, improve collaboration, and ensure stronger compliance and quality control.
Key Metrics & Results
50%
FASTER
Document approvals
70%
LESS
Time spent on audit preparation
100%
GLOBAL
Standardization of eSign workflows
GxP Use Cases via MSB Docs
eSignature
- Investigation Plans, RCIR, Interim & Final Investigation Reports
- Accelerates root cause analysis and timely deviation closure
- IQ / OQ / PQ, Release Certificates, RP Checklists
- Enables faster batch readiness and manufacturing approvals
- Summary Reports, Hypothesis Reports, Study Protocols
- Supports management review and regulatory submissions
- Data Mapping Checklists, Traceability Sheets
- Strengthens data integrity and audit confidence
- Training materials, attendance sheets, acknowledgements
- Ensures workforce compliance and inspection readiness
- Change summaries, justifications, errata
- Enables controlled, compliant change management
- Validation certificates, FDA forms, re-injection recommendations
- Provides formal, and inspection-ready compliance artifacts
- Annexure IX, FMEA forms
- Supports structured risk evaluation linked to quality events
Conclusion
By implementing MSB Docs’ 21 CFR Part 11 compliant eSignature solution with seamless API integrations, Glenmark successfully digitized and standardized its global documentation workflows. This transformation eliminated manual inefficiencies, strengthened compliance and governance, and enabled faster, audit-ready operations across all critical GxP processes.



